|
|
|
|
Family: Frullaniaceae
[Frullania aterrima var. aterrima autonym, moreFrullania weymouthiana Steph., Jungermannia aterrima Hook.f. et Taylor] |
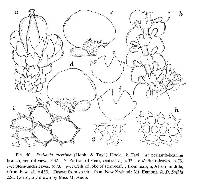
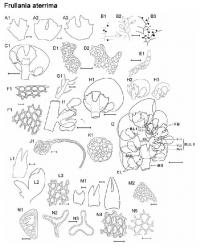
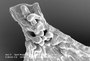
Detailed description: Plants small to medium (main shoots to 1000 µm wide), forming olive-green, copper-brown, to black patches, closely to loosely adhering to substrate. Leading stem to 10–30 mm long, and up to 125µm in diameter, 6–9 cells wide, little differentiation between cortical cells (14–26 in no.) and medullary cells (16–30 in no.), former slightly smaller than the latter, both with firm walls, lumen irregularly shaped. Branching often regularly pinnate (primary branches to 15mm long) to bipinnate (secondary branches to 8mm long), occasionally tripinnate (tertiary branches to 3mm long) in larger plants, branches usually with progressively smaller leaves. Dimorphic branching, with both Frullania-type (FB) and occasionally, Lejeunea-type (LB) branching. Initial appendages of FB: First branch underleaf (BUL1) always with three distinct segments, the ventral lamina divided for 1/3–2/3 its length into two unequally or subequally sized lobes + 1 dorsal saccate lobe; First branch leaf (BL1)usually± characteristic of normal stem leaves (i.e.BL1: 1 explanate dorsal lobe + 1 saccate lobule + 1 stylus), only rarely are the appendages of BL1 reduced in size and elobulate with leaves characteristic of the main stem starting at BL2–3. Initial appendages of LB: BUL1 and BL1–3 are always reduced in size and lobule-free; formation of normal lobulate leaves occurring thereafter. Stemleavesof main branchflat when dry and wet, slightly imbricate to contiguous, suborbicular to broadly oval, to 700µm long × 550µm wide with incurved distal margins, and dorsal margins extending beyond the farther edge of the stem, rounded apices and non-auriculate at the base, margins entire, dorsal surface smooth. Lobules ± remote from the stem (lobule attached to stem by (2) 3–4(5) cells) and at angles of (20) 30–50 (60)° with the stem, thus the lobules normally tilted outwards; cylindrically pitcher-shaped (orbicular in cross-section with up to 30 cells in circumference) and 2–2.5× long as wide; lobules large (its area obscuring ¼–⅓× the exposed area of the dorsal lobe), (150) 175–280 (300) µm long × (60) 75–120 (130) µm wide; lobules often darker in colour than other organs (in part due to heavily pigmented cell walls);somewhat dorsiventrally compressed near mouth as compared to gibbous upper half, the opening wide, extending along the abaxial lobule margin; immediately above the lobule mouth there is usually a ± discoloured, gibbous, protuberant cell; free margin of lobular mouth crenulate-sinuate, usually hyaline near mouth, lobule apex obtuse. Stylus small, usually uniseriate or occasionally subtriangular, up to 50 µm long, typically 2–3 (4) cells wide at base + 2–4 (6) cells often forming an arched filament, the stylus with the tip uniseriate for 2–3 (4) cells, usually terminating in a slime papilla ((4) 5–10 (12) cells in total). Underleaves of leading stems ⅓ to ½ the size of leaf lobes, often distant to contiguous or just overlapping with lobules, underleaves contiguous to distant from each other, often slightly broader than long, occasionally long as wide, ca. 140–240 µm long × 120–250 µm wide, broadest at middle (to 32 cells wide); 1–2 (3) pair/s of blunt-acute teeth or angulations produced on free lateral margins (may be absent on underleaves of secondary branches); teeth largest at middle of underleaf, to 30µm long, triangular, acute apex, 3–7 cells in total, teeth and/or angulations towards apex and base of underleaf becoming reduced in size, to 10 µm long, and 1–4 cells in total; apex of underleaf bilobed usually to ½ its length, lobes separated by a narrowly U to V-shaped sinus, occasionally a minute tooth on free margin of sinus, lobes with acute to subacute apices and to 15 cells wide at base. Rhizoid-initial area present near base of underleaf, rhizoids often seen, subhyaline, in short bundles. Not strictly microphyllous, lobules of secondary stems ± similar size to main stem, but lobes and underleaves of secondary branches markedly smaller than those of leading stems. Underleaves of secondary branches (at least in smaller plants) also less conspicuously toothed, or teeth otherwise absent, when toothed often with only one pair of teeth on the free margins and often with a V-shaped sinus separating the underleaf lobes. Lobe marginal cells ± rectangular, hyaline walls subequally thickened, cell cavities brownish red, 8–10 µm long × 7–9 µm wide; median cells ± subquadrate to rectangular, hyaline walls subequally thickened, intermediate thickening rare to absent, wall thickness to 2.5 µm wide (without intermediate thickenings), cell cavities of median cells brownish red, (10) 12–18 (20) µm long × (9) 10–14 (15) µm wide (1–1.75× long as wide); cells becoming gradually larger basally, cavities of the basal median group of cells (30) 38–50 (55) µm long × (22) 25–32 (35) µm wide; walls of basal cells with subtriangular trigones and intermediate thickenings, walls and cavities brownish red. Median cells of underleaves ± subequally thickened so that the hyaline trigones and intermediate thickenings become indistinct, cell cavities to 15 µm long × 10 µm wide. Median cells oflobuleslightly longer than wide, cell cavities to 14 µm long × 8 µm wide, walls flexuose with indistinct trigones and small nodulose intermediate thickenings (both often heavily pigmented, olive-brown to dark brown in contrast to hyaline walls of the lobe and stem underleaf). Oil-bodies of the leaf lobe median cells: 2–3 (4) per cell, typically small, spherical (1–3 (4) µm in diam.), to ovoid or ellipsoidal (2–5 (6) × 1–4.0 (5) µm), subhyaline, without any significant, visible, internal structure i.e. giving the appearance of being almost homogeneous. Usually a conspicuous group of 2–4 basal cells each with a single, large (to 35 µm long × 20 µm wide), grayish, fine-coarsely granular oil-body (ocellus) that almost obscures the entire cell lumen. The oil-bodies of the median cells are often similar in size to or slightly smaller than the chloroplasts; occasionally the oilbodies are larger than the chloroplasts. The oil-bodies of the lobule and underleaf are similar to those encountered in the leaf lobe. Asexual reproduction;none known Dioicous (rarely some phenotypes monoicous). Androecia subspherical to discoid, 450 µm long ×350 µm wide, 2–4 (6) pairs of bracts, sessile or terminal on very short-stalked branches (stalk rarely leafless, usually with 1–4 vegetative leaf lobes). Gynoecia terminal on main or leading stem. A shoot system usually arising immediately below the set of 3–4 progressively larger bracts below the gynoecium or perianth (subfloral branch), occasionally arising one or more complete-leaf cycles below the gynoecia or perianth bracts i.e. a subfloral branch, or rarely occurring between bracts and perianth (subfloral innovation). ♀ bracts and bracteoles usually in 3–4 pairs, gradually increasing in size and complexity, distally, towards the gynoecium; or occasionally in 2 close overlapping cycles. Innermost bract unequally bilobed; bract-lobe ovate to oblong, to 625 µm long × 300 µm wide, tapering to a subacute to almost appendiculate apex, the margin entire, bract-lobule shorter and narrower, ovate–lanceolate, 300 µm long × 175 µm wide at base, subacute to rounded apices, with a small stylar tooth at the base 3–4 cells in size; innermost bracteole free from bracts, ovate to oblong, to 300 µm long × 250 µm wide, ⅓–½ bilobed with subacute lobes and V-shaped to U-shaped sinus, entire margins. Marginal cells of bract and bracteole ± subequally thickened, but towards the median cells, trigones becoming large and bulging. Two archegonia per gynoecium. Perianth freely emergent, to 1250 µm long (excluding beak) × 900 µm wide, exterior smoothly trigonous, oblong-ovate, tapering towards the apex into a short beak; perianth beak cylindrical, to 90 µm long × 70 µm wide, expanding slightly only at the smooth rim of mouth; the inner beak surface densely covered with large single-celled protuberances, to 25 µm long (occasionally protruding to the exterior of the perianth mouth). Median cells of perianth with large bulging, convex trigones, intermediate thickening rare or absent. Seta to 1.2 mm long and 6–8 (9) cells in diameter, with up to 26 epidermal cells and 24 medullary cells (below the hypophysis); valves of capsule to 500 µm long × 250 µm wide; elaters (28) 32–42 (44) in total (common arrangement on two alternating valves: 2+4+3; 1+3+5, elaters rarely arranged in rows of 4 on the valve); unispiral, 220µm long × 20 µm wide, elater surface irregularly rugose-granulate. Epidermal cell layer (11) 13–17 (19) cells at widest region, epidermal cells typically polygonal in outline, the thickenings at the angles of the epidermal cells do not extend out as lobes; consequently the juxtaposed thickenings of 3–4 adjoining corners form a configuration with an evenly rounded or oval outline. Inner cell layer: portions of the radial walls are spread irregularly over the free tangential face and are strongly thickened, forming a sinuate partial layer of thickening or jig-saw like pattern; cells often elevated into ± obtuse keels, juxtaposed to form ridges arranged longitudinally and parallel with the long valve axis and as hexagonal rings. Surface ultrastructure of the inner cell layer densely covered with fine perforations. Spores globose, to 48 µm at the widest axis, and interspersed with 5–9 rosettes; each rosette 4–7.5 µm in diam., bearing a ring of 6–8 conspicuous protuberances; these primary projections 1–3 µm long × 0.75–2 µm wide at base, usually with a 2.5–3:1 length to width ratio (i.e. projections typically longer than wide), and taper gradually to a subacute apex, occasionally with 1-2 simple short branches, and occasionally pappilate - at most with 1–2 minute secondary deposits (radially symmetrical, low protuberances); spore wall papillae densely distributed between areas of the rosettes. Etymology: The type specimen is a blackish plant and presumably it is named after this feature (from Latin aterr meaning black). Affinities, differentiation & variation: This species is allied to other species of sect. Aterrimae, all sharing a suite of features, including 1) toothed underleaves (at least those of the main stem or leading branches); 2) a small and inconspicuous stylus, usually with an arched filament, terminating in a unicellular row of 2–3 cells; and 3) a conspicuous group of basal ocelli in the leaf lobe. Frullania aterrima can usually be distinguished from other species of sect. Aterrimae by the plant being dioicous and by the first branch leaf, which is usually lobulate and inflated; other species of this section are typically monoicous and have elobulate first (and often) second branch leaves. Furthermore, under the SEM, the projections forming the rosette pattern on the surface of the spores of F. aterrima lack any significant secondary surface ornamentation. In contrast, the other species of sect. Aterrimae have projections with varying degrees of secondary surface ornamentation. Monoicous forms of F. aterrima have been encountered at higher altitudes or colder than usual habitats, but these forms are otherwise morphologically identical to F. aterrima s. str. In these cases, the inflated lobule of the first branch leaf is often a good character to separate this species from other species of sect. Aterrimae . Frullania aterrima can immediately be distinguished from F. hattorii by the smooth dorsal lobe surface and subsequent absence of any surface ornamentation of the leaf lobes; it can also be separated out from F. colliculosus by the distinctly 3-keeled perianth, in contrast to the plicate perianth with a colliculate surface for F. colliculosus. A range of variation in the development of plant size, and the form of lobes, lobules and underleaves, occasionally, may offer confusion with other Frullania species. For instance, small plants of F. aterrima may be mistaken for the small species, F. subrostrata, but the latter differs in the explanate lobules of the first and usually the second branch leaf, the perianths dorsally with a deep and narrow longitudinal sulcus, and the plants are always monoicous with gametangia usually present. The variation in underleaf dentition ranges from underleaves with 1, 2 or occasionally three very prominent teeth on the lateral margins, to underleaves that are barely toothed and the teeth may be reduced to only a strong angulation. In these cases, F. aterrima is most apt to be confused with phenotypes of F. rostrata that also have angulations or 1 very weak tooth present, but the latter species differs by: the absence of basal ocelli of the leaf lobes; the larger, more conspicuous stylus; the lobules rarely at angles greater than 50° to the stem; and the uniform colour throughout the plant. Furthermore, chemically these two species are quite distinct from each other with F. aterrima having different sesquiterpene lactones from F. rostrata; in addition to a diterpene hydrocarbon, kaurene, which is not a major chemical constituent of F. rostrata. In New Zealand, some phenotypes of F. aterrima may superficially resemble some forms of F. ptychantha from subg. Diastaloba. In this situation, F. aterrima has secondary branches withthe leaf lobes and underleaves dramatically smaller than those of the main stem, and the branches subsequently appear as microphyllous branching - a feature that is typically associated with F. ptychantha. Furthermore, in F. aterrima, the lobules of the secondary branches may be almost horizontal to the main stem, which is also a feature of F. ptychantha. However, F. aterrima is easily differentiated from F. ptychantha by the presence of basal ocelli, the toothed underleaves, the lobules usually at least 1.5× long as they are wide, and only a single saccate lobule at the first branch leaf.
Field recognition: The typical form of this species may be readily recognised in the field by the combination of the following features: the olive-black colour of the plant; the glossy black 3-keeled perianth; and when held up to the light, the lobules at strong angles in relationship to the stem coupled with the darker colour of the lobules in relation to other organs. Geographic distribution: New Zealand: throughout the North Island, South Island, Stewart Island and the sub Antarctic islands. Australia: Victoria, Tasmania. At least in New Zealand, Frullania aterrima is particularly abundant in cooler, exposed habitats, e.g., in regenerating scrub of Leptospermum scoparium (manuka) on the frost flats of the central North Island and in stunted Olearia colensoi (leatherwood) forests at higher elevations. It is also interesting to note that some of the phenotypes outlined above may possibly be the product of hybridisation and this possibility requires further analysis. In all cases, when phenotypes of F. aterrima resembled the form of other species, for example, F. rostrata and F. ptychantha, those species were growing with F. aterrima. Hodgson (1949) also noted a specimen that appeared similar to F. ptychantha and commented that it could well have been the result of a cross between F. aterrima and F. ptychantha.
Plants small to medium in size and typically monoicous; leaf lobes smooth or papillate; perianths 3-keeled or plicate, smooth or with the surface cells raised as hill-like elevations; initial branching appendages typically of the Rostrata-type and Chevalieri-type. Notes¾All species of this subsection are typically monoicous and have distinct spore wall sculpturing within subg. Microfrullania. The rosette-forming projections that have secondary short branches and conspicuous minute secondary deposits, radially symmetrical, low, protuberances; F. hattorii is distinctive with the cells of the dorsal lobe having conspicuous projections. Species of this subsection are possibly derived from F. aterrima. |